A 48-year-old attorney comes in after his second stroke in eighteen months. Blood pressure is fine. Cholesterol is fine. He has never smoked. The imaging shows deep white matter changes and small lacunar infarcts in a pattern that does not match any of the usual vascular suspects. His father died of "early dementia" at 62. His aunt had three strokes before 55. The cardiologist has already cleared him. The primary care doctor is out of ideas.
This is CADASIL until proven otherwise.
CADASIL stands for cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy. It is the most common inherited cause of small vessel disease in the brain, and it is also one of the most underdiagnosed. A mutation in the NOTCH3 gene produces a defective receptor on the smooth muscle cells and pericytes that line small cerebral arteries. Those cells fail. The vessels scar from the inside. Granular osmiophilic material accumulates in the vessel walls. Over years, the brain's deep white matter is starved of reliable blood flow.
What CADASIL Actually Looks Like
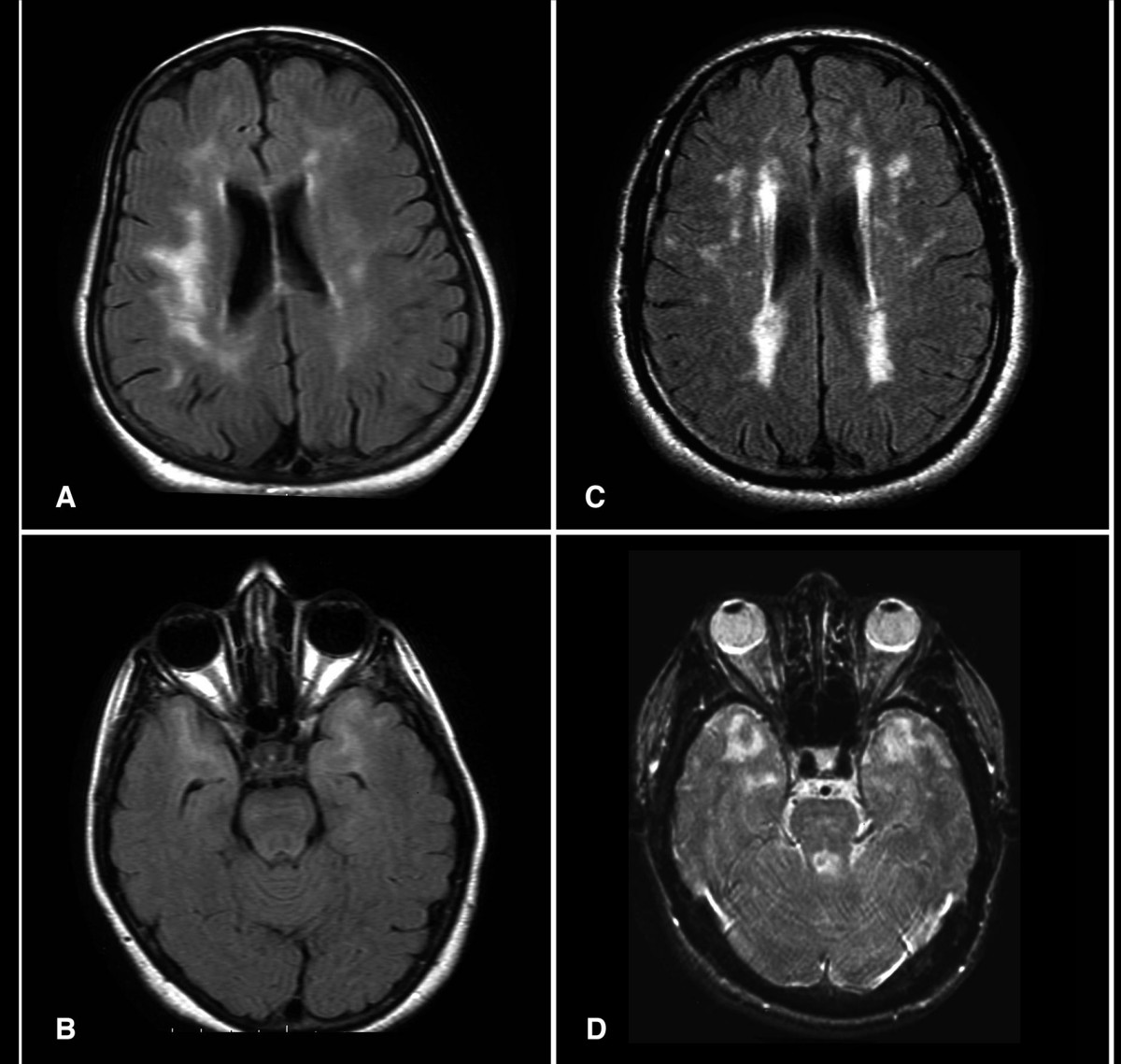
The classic phenotype unfolds in a recognizable sequence. Migraine with aura appears first, often in the third decade. Roughly 30% of patients describe it, and the aura is frequently atypical or prolonged. The stroke chapter opens later, with a mean onset around age 49 and a range that stretches from the mid-twenties to the seventies. These are subcortical ischemic strokes, and they arrive without the hypertension, diabetes, or tobacco history that usually precedes them.
Cognitive decline is the third act. Early changes target executive function and attention rather than memory, which is why patients and families often miss it. The forgetfulness that rings alarm bells in Alzheimer's disease is relatively spared in CADASIL. Instead, the patient loses the ability to plan, organize, and sustain focus. After age 60, roughly 60% of patients progress to multidomain dementia. Depression, apathy, and adjustment disorders show up in about a third of patients and are often treatment-resistant.
The disease lives in the central nervous system. It does not involve the kidneys, the heart, or the peripheral nerves, which is one of the features that distinguishes it from systemic vasculitides and hypercoagulable states. On MRI, white matter hyperintensities are the earliest and most consistent sign, often visible decades before the first stroke. Over time, lacunes accumulate, microbleeds appear, and the brain begins to atrophy.
The Genetics Are More Complicated Than Textbooks Suggest
CADASIL is caused by cysteine-altering mutations in the NOTCH3 gene, which encodes a receptor with 34 epidermal growth factor repeat domains. The classic mutations either add or remove a cysteine residue within those repeats. Where the mutation sits in the gene matters clinically. Variants in the first six repeats and in repeats 18 through 34 are associated with earlier stroke onset, a higher burden of encephalopathy, and a greater risk of dementia.
The older estimate for CADASIL prevalence was 2 to 5 cases per 100,000 people. Newer data from a 2025 JAMA Neurology staging study by Gravesteijn and colleagues challenge that number. When researchers looked at cysteine-altering NOTCH3 variants in population databases, they found them at frequencies of 2 to 10 per 1,000 individuals. That is a two-to-three-orders-of-magnitude difference. What it means is that the CADASIL phenotype is a spectrum. Severe familial cases sit on one end. Milder, later-onset presentations that look like sporadic small vessel disease sit on the other. Many of these milder cases are never genetically diagnosed because no one thinks to test.
The same 2025 paper proposed a staging system for NOTCH3-associated small vessel disease based on imaging burden and clinical features rather than just the classic familial phenotype. Applying modern staging requires the kind of advanced imaging and longitudinal cognitive tracking that a precision neurology workup is designed to provide.
The Differential Is Crowded
CADASIL is the most common inherited small vessel disease, but it is not the only one. A short list of its cousins is worth knowing.
CARASIL, the autosomal recessive version, is caused by HTRA1 mutations and typically presents between ages 10 and 30. Its triad of backache, baldness, and brain attack reflects systemic involvement that CADASIL lacks. A dominant HTRA1 variant also exists, with a milder and older-onset phenotype.
Fabry disease is an X-linked lysosomal storage disorder caused by alpha-galactosidase A deficiency. It can cause stroke, but it also produces kidney disease, cardiomyopathy, angiokeratomas, and painful peripheral neuropathy. Recognizing Fabry matters more than most differentials because enzyme replacement therapy is available.
CARASAL, from CTSA mutations, presents with prominent brainstem symptoms: tinnitus, hearing loss, and dysphagia. RVCL-S, from TREX1 mutations, includes retinal vasculopathy along with liver and kidney involvement. COL4A1 and COL4A2 mutations produce a spectrum that includes HANAC syndrome and PADMAL. MELAS, the mitochondrial stroke syndrome, brings muscle weakness, recurrent vomiting, and stroke-like episodes. Each of these has a pattern that, once recognized, is hard to miss. The problem is that no one is looking.
Why the Diagnosis Matters Even Without a Cure
No disease-modifying therapy exists for CADASIL. That is the hard truth. The case for diagnosis is not that treatment reverses the course. The case is that diagnosis changes everything else.
Hypertension in a CADASIL patient is a far greater threat than it is in the general population, because the vessels are already structurally compromised. Tight blood pressure control reduces the risk of incident dementia, disability, and recurrent stroke. Smoking is independently associated with earlier stroke onset and needs to be addressed directly. Donepezil offers modest benefit for executive function, though not for overall cognition. SSRIs are a first-line option for the depression and apathy that often dominate the clinical picture, even when response is only partial.
Diagnosis also reframes the family. CADASIL is autosomal dominant, which means each first-degree relative has a 50% chance of carrying the variant. Genetic counseling becomes part of the care plan, not a footnote to it. Imaging surveillance in at-risk relatives can catch white matter changes decades before symptoms. Lifestyle and vascular risk factor management becomes preventive medicine with actual teeth.
This is the kind of problem our Intensive Brain Health Program is built around. High-resolution imaging, biomarker workups that look beyond the standard vascular panel, genetic testing when the clinical pattern warrants it, and a care model that tracks cognition over years rather than minutes. The patients most likely to benefit are the ones who have already been told nothing is wrong.
When migraine with aura in a 30-year-old sits alongside an uncle with early strokes and a grandfather who died of early dementia, the family pattern is the diagnosis waiting to be made. The answer is not always in the standard panel. Sometimes it is in the gene.
Dr. Sean C. Orr, M.D., is the founder of The Neurogenesis Project, a brain health clinic specializing in precision neurology, cognitive optimization, and the treatment of neurodegenerative conditions.